Unicellular Organisms – The Cambrian Explosion – Nano Machines
Unicellular Organisms – The Cambrian Explosion – Nano Machines
So Many Moving Parts –How Do They Fit?
By Ralph D Zehr M.D.
(Presented at the 2016 Science Symposium)
Unicellular Organisms
It is widely assumed that life began as a unicellular organism. An understanding of cellular morphology and function, particularly in unicellular organisms, is therefore essential to any serious study of the origin of life. All living cells belong in one of two groups, prokaryotes and eukaryotes.
Prokaryotes appear to be much simpler, are devoid of most organelles except for ribosomes. They do contain an area where DNA molecules are relatively concentrated in the central region of the cell, in contrast to eukaryote, in which the intracellular DNA is enclosed in a nuclear membrane or envelope.
E. coli, have been by far the most extensively studied prokaryote because of their simplicity, and rate and ease with which they reproduce. Their complete genome has been deciphered. Though one might have expected to find far simpler proteins and less complex intermolecular interactions than those observed in eukaryotes, that has not proven to be the case. Electron microscopy has revealed a relatively large collection of DNA molecules in an area termed the nucleoid, located at or near the center of the organism, as well as many small ribosomes in the cytosol. Complex interactive processes between DNA, mRNA, and protein synthesis occur. E. coli have multiple flagella providing impressive mobility. They can react to their environment by moving toward nutrient sources, as well as slowing their metabolic rate when nutrients are scarce. They can reproduce rapidly, undergoing cell division every 20 min. under ideal circumstances. A properly prepared culture media, inoculated with E. coli, when incubated overnight can yield millions of identical living organisms ready for study the following morning.
One can hardly mention the role that E. coli has played in research without recognizing how it contributed to elucidating the “proton – motive force" and the associated chemiosmotic hypothesis, proposed by Peter Mitchell in 1961. He recognized a linkage between electron transfer and adenosine triphosphate (ATP) synthase generation of ATP. He was convinced there was an electrical phenomena associated with the chemical process. There was an electron transfer process, where electrons pass down an electron gradient, along a chain of several molecules across or along cell membranes, yielding their energy in small increments as they pass. As this proceeds, protons accumulate outside the membrane, building a proton gradient, known as the “proton – motive force.” They subsequently pass down their gradient, driving machines such as ATP synthase. It actually required some 16 years for his theory to be widely accepted. In 1978 Mitchell was awarded the Nobel Prize in chemistry. Not only did his hypothesis of chemiosmotic become well established but it was also discovered that this phenomena occurs in many bacteria, in all animal mitochondria and in chloroplasts throughout the plant kingdom. (1)
Another example of what we can expect from further study of prokaryotes, is the recent documentation of aquaporin in E. coli, which is highly specific and displays a rapid rate of water flow through its channel. It is expected to provide a useful model for further study of aquaporins which are large complex macromolecular proteins located in cell membranes and in general are responsible for controlling the flow of water and glycerol in and out of cells. (2)
Even though prokaryotes display relatively few subcellular structures, there appears to be significant subcellular organization or intracellular compartmentalization on a molecular level. Microbiologists, Lucy Shapiro and Richard Losick, stated, "The use of immunogold electron microscopy and fluorescence microscopy to study the sub-cellular organization of bacterial cells has revealed a surprising extent of protein compartmentalization and localization." They go on to describe such examples as DNA polymerase, cell division proteins, and bacterial cytoskeleton. (3)
Eukaryotes populate the bodies of the entire animal kingdom and represent our general concept of a typical cell. They display extensive intracellular compartmentalization with a nucleus surrounded by a galaxy of highly sophisticated organelles performing specific functions. Their energy systems are well-defined and highly specialized. Chloroplasts in plants function as solar panels, transforming the energy of the visible spectrum of sunlight into chemical energy in the form of sugars, starches, cellulose and ATP, whereas, animal mitochondria transform the stored chemical energy in the ingested sugars and starches, as well as fatty acids, into ATP. Energy can also be extracted from unneeded ingested proteins and amino acids if the diet contains an overabundance.
Franklin M. Harold , professor emeritus of biochemistry at Colorado State University, whose professional career spanned 40 years of research focused on microorganisms, provides this interesting perspective on our understanding of life based on our knowledge of cells. In his recent book The Way of the Cell, he stated, "Biochemists insist, rightly, that when one takes cells apart one finds nothing but molecules: no forces unique to life, no cosmic plan, only molecules whose writhings and couplings underlie and explain all that the cell does. Thus Max Perutz, reflecting on the mechanisms that allow E. coli to detect and swim towards a source of nutrients, found nothing that could not be ‘reduced to chemistry.’ I share the commitment to a material conception of life, but that makes it doubly necessary to remember that before the cells were taken apart – as long, in deed, as they were alive – they displayed capacities that go beyond chemistry. Homeostasis, purposeful behavior, reproduction, morphogenesis, and descent with modification are not part of the vocabulary of chemistry but point to higher levels of order. Even as a catalog of small parts approaches completion, the transition from molecular chemistry to the supramolecular order of cell emerges as a prodigious challenge to the imagination. Make no mistake about it: here we touch, if not the very secret of life, at least an essential stratum of that many layered mystery. For if life is to be convincingly explained in terms of matter and energy, organization is all that stands between a soup of chemicals and a living cell.” (4) (Added emphasis is mine.)
The Cambrian Explosion: How Does It Fit?
The expected rate for new phyla to arise de novo, as predicted by Darwin's theory, would follow a relatively smooth logarithmic or exponential curve over time. As a result of small incremental changes in size and complexity of living organisms based on a long series of random mutations of the genetic code, followed by intervening periods of trial and error resulting in natural selection of the fittest, one would expect to find a trail of fossils displaying incremental changes of primitive phyla, starting with a simple organization and body plan, leading through many progressive stages to more complex morphological arrangements of the body parts, characteristic of more advanced phyla. One would then expect to see a continuous protracted spectrum of new phyla spread over eons of time, displaying a branching pattern as new forms branch off randomly in different directions.
But this is not what happened based on the fossil records. During the Cambrian Explosion as recorded in both the initially studied fossil deposits in the Burgess Shale in eastern British Columbia, Canada, discovered by Charles Doolittle Walcott in 1909, and in the more recently studied most exquisitely preserved and more detailed Cambrian period fossils found in China near Chengjiang, a very different story emerges. Both deposits, located nearly halfway around the world from each other, show a surprisingly similar picture of a Biological Big Bang (5),(6) in which many phyla and recognized body plans, made their appearance during an instant of geological time representing approximately 0.1% of the Earth's total geological history.
But, why do we not find fossil evidence of simpler precursor organisms showing a gradual progression of complexity leading to each individual unique body plan characteristic of each phyla? In fact, there is very little evidence of any intermediate forms of any of the phyla. The identified phyla display considerable morphological stability. In other words, we are unable to isolate fossils that show significant evidence of simpler organisms gradually progressing in complexity leading to the more advanced body plan of the phyla observed. Nor do we find fossil records in either a specific geological time or over all of geological history in which there is morphological evidence of a long series of gradual progressive stages linking the phyla or confirming an evolutionary progression from simpler organisms.
A discussion of all the phyla identified in the Cambrian fossil deposits is beyond the scope of this paper but a brief listing and discussion of the most familiar, seems appropriate. These include: Brachiopoda, Eldoniaioda, Annelida, Ctenophora, Hyolitha, Chordata, and Arthrodada.
Chordata, characterized by the presence of a notochord, includes all vertebrates. An interesting finding in the Chengjiang fossil deposit is the presence of all three subphyla of Chordata, Cephochordata, Craniata, and Urochordata.
In his book, Wonderful Life, The Burgess Shale and the Nature of History, Stephen Jay Gould raises two questions related to the Cambrian Explosion: ”(1) Why did multicellular life appear so late? (2) Why do these anatomically complex creatures have no direct, simple or precursors in the fossil record of Precambrian times?” (7)
The Cambrian period fossil deposits in China display most exquisitely the fine details even of many soft tissue parts: "The lower Cambrian Sediments near Chengjiang have preserved fossils of such excellent quality that soft tissues and organs, such as eyes, intestines, stomachs, digestive glands, sensory organs, epidermis, bristles, mouths, and nerves, can be observed in detail. Even fossilized embryos of sponges are present in the Precambrian strata near Chengjiang. (8)
“Cambrian-level strata show soft body parts of jellyfish-like organisms (known as Eldonia) such as radiating water canals and nerve rings. These fossils even include the gut contents of several different kinds of animals and undigested food residue in their stools.” (9)
Recent work performed by Michael S. Y. Lee and colleagues at the University of Adelaide, compared the relative rates of evolutionary morphological development and molecular development during the Cambrian Explosion, to that since the Cambrian period up to modern times. They chose to study arthropods since they are such a large and diverse group of living animals during the Cambrian period as well as since. "Using 395 morphological characters, 62 protein coding genes, and 20 calibration points from the fossil record, the authors inferred the evolutionary histories using a number of different analytical methods, and their relationship results are consistent with other recent research." (10) They concluded that the evolutionary rate of morphological change during the Cambrian period was four times the average rate since the Cambrian period, while the average estimated rate of molecular evolution during the Cambrian period was five times the average rate since that time. (11) (See figures 1 and 2 below.)
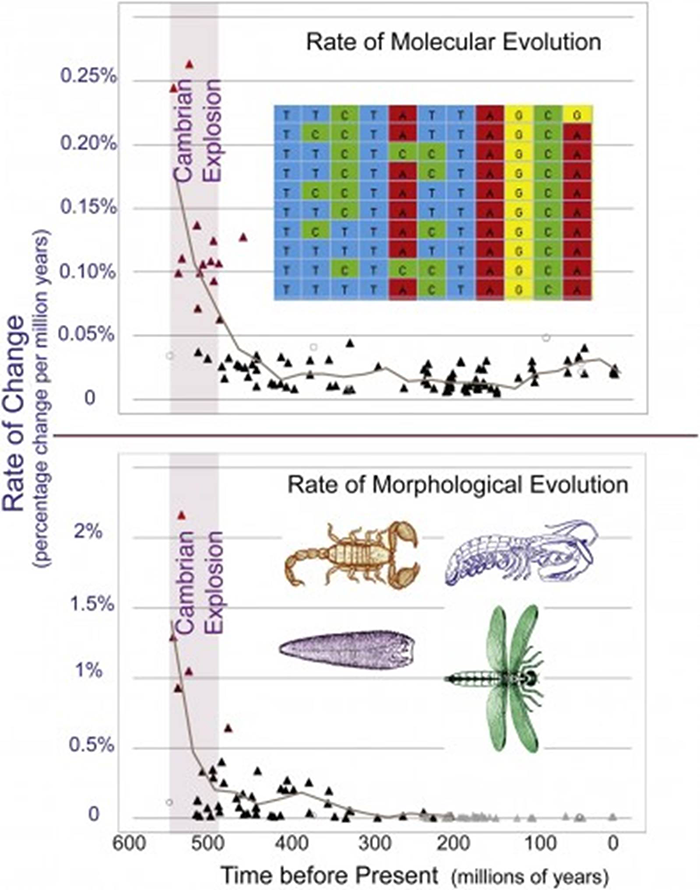
Rates of morphological and molecular evolution in arthropods increased 4 to 5 times during the Cambrian Explosion. Courtesy of Current Biology. Figures one and two. (11)
It is most fortunate to have found a fossil bed yielding such superbly preserved specimens in which many of the soft parts can be clearly identified and fine detailed morphology is preserved. The informational value of these fossil beds is multiplied because of the illumination they provide on such a critical and unique period in the evolutionary history of life on our planet. If paleontologists were enthralled with finding the Burgess Shale fossils, it can be reasonably assumed that they were ecstatic with the discovery of the Chengjiang fossil bed. In general, organisms without skeletal structures rarely leave fossil records of their existence. In the case of the Cambrian Explosion, we find that the pages of the great stone book of geological history covering the Cambrian Explosion, are among the best preserved in the entire book.
During Darwin's time, accurate dating of rocks by radiometric means was unavailable. Fossils were sorted based on phylogenetic relationships. At the time of the discovery of the Burgess Shale fossil deposits, the best estimate of their age was during the Cambrian Period, thought to have begun approximately 570 million years ago and ended 510 million years ago. This provided a window of 30 to 60 million years for the Burgess Shale fossils to be formed. In 1993 a Cambrian deposit in Siberia was identified containing zircon crystals in close proximity, just above and just below. Based on radiometric dating these were found to be 525 million and 530 million years old. The highly precise measurements narrow the window of deposition of fossils establishing the Cambrian Explosion of living organisms to an instant in geological time. Placed in perspective of a 24-hour day representing the estimated total history of life on earth, estimated at approximately three billion years, the Cambrian Explosion occupied about one-half minute early in the first hour of multicellular life on our planet.
This scenario has little resemblance to the process described by Darwin in which he envisioned living organisms displaying an extended series of numerous, slight modifications over a long time period, resulting eventually in new body plans that were better adapted to the environment and more capable of survival.
Another unexpected factor that characterizes the organisms depicted by the Cambrian Explosion is the apparent sudden appearance of representative organisms of most, of the phyla, seemingly coexisting together. The phyla represent the highest levels of the divisions of animal life as we understand them. In an instant of geological time, most if not all of the major divisions of animal life made their appearance. Instead of a bottom up development of the simplest life forms, driven by random genetic mutations causing numerous, small alterations in morphology and physiology that proved beneficial to their survival, eventually leading to multiple large distinct divisions of living organisms, we observe an apparently top-down development in which these major divisions of animal life display a large portion of body plans ever to appear on Earth. One might conclude that the Cambrian Explosion depicts the "origin of phyla” rather than “origin of species.” The origin of the species would come much later following extensive sifting and sorting of the basic life forms, eventually leading to the phenomenal diversity of living organisms that we all observe everywhere we look.
As time passed, the basic dilemma facing Darwinism has shifted. Again Gould summarizes the situation succinctly, "Darwin has been vindicated by a rich Precambrian record, all discovered in the past thirty years. Yet the peculiar character of this evidence has not matched Darwin's prediction of the continuous rise in complexity toward Cambrian life, and the problem of the Cambrian explosion has remained as stubborn as ever – if not more so, since our confusion now rests on knowledge, rather than ignorance, about the nature of Precambrian life." (12)
What Are the Foundational Building Blocks of Living Organisms?
Atoms and molecules are the building materials from which all living organisms are made. All living organisms are dependent on thousands of chemical reactions that occur continuously and simultaneously throughout their lifetimes. They are exquisitely coordinated, constantly modified and controlled by the central nervous system, continuously responsive to feedback from internal stimuli, adjust to external environmental changes, are directed by thousands of genetic instructions, and greatly influenced by carefully balanced hormonal influences. All of these factors controlling and directing living creatures are based on chemical interactions within the living organism. How the limited number of different elements that compose the many trillions of molecules that carry out these incredibly complex processes in all living organisms, can achieve such diverse functions and at same time maintain the stability and functional predictability so essential to life, remains an unsolved mystery.
Another interesting consideration is the fact that the very material of which living organisms are composed, is constantly being replaced by other similar atoms. It is difficult to estimate the exact turnover rate of many of the atoms that make up our body parts since this varies greatly from one tissue to another, and from organ to organ, however, on average they are replaced many times over, during a lifetime. We know that the proteins within a given cell are constantly built up and torn down and the basic amino acids are constantly recycled. Whole cells are also constantly replaced by younger ones. Our red blood cells are recycled every 60 to 90 days and the entire endothelial lining of the small intestine, responsible for absorption of the nutrients we require, is recycled every five days on average. (13)
Types of Chemical Bonds which Hold the Parts Together
In living organisms the atoms are held together by a series of different types of chemical bonds of varying strengths.
Covalent bonds are by far the strongest chemical bonds. Covalent bonds form when there is exchange or sharing of one or more electrons in the outer shells of two or more separate atoms. They may be single, double, or triple. The most common double covalent bonds are between carbon and oxygen, carbon and nitrogen, carbon and carbon, and phosphorus and oxygen. The covalent bond between phosphorus and oxygen is of particular interest since it is the inherent energy in this bond that is utilized to transfer, distribute, and energize essentially all physiological functions throughout the animal kingdom in the form of ATP.
There is a group of noncovalent bonds that are much less energetic but equally important. The non-covalent bonds are important in stabilizing macromolecules, particularly maintaining their exact folded configuration. Of these, hydrogen bonds are of great importance. They form primarily between hydrogen and oxygen and hydrogen and nitrogen. They are strongest when positioned in a straight line, however very frequently nonlinear angular hydrogen bonds contribute more effectively to the stabilization of the three-dimensional configuration, known as conformation, of large protein molecules. The precise folding of the long chains of amino acids making up many macromolecular proteins is critical to their proper functioning. The same protein compound can have very different functions depending on how it is folded.
Hydrogen bonds are extremely prevalent in water, both when containing dissolved Ions and in pure water. Hydrogen bonds are generally about double the length of covalent bonds between the same elements. The covalent bond length between hydrogen and oxygen in water is approximately thirteen nanometers (nm), whereas the hydrogen bond between hydrogen and oxygen in water measures approximately twenty-seven nm in length. These are average lengths since the bonds are constantly changing in length. See figure 3 below. (One nm is one billionth of a meter.)
Van der Waals interactions are nonspecific and occur between all atoms in close proximity. They vary in strength depending on the distance between the nuclei of the adjacent atoms. The minimal distance is limited by the van der Waals’ radius which is equal to the radius of the atomic sphere occupied by a given nucleus and its surrounding associated electron cloud. The van der Waals attractive force increases until it reaches a maximum at which point the repulsion of the overlapping negatively charged electron clouds of the two atoms balance the attractive force. This is known as the van der Waals contact. The strength of this attraction is small, measuring in the range of 1 kcal per mol. (One kcal equals one thousand calories.)
Another weak nonspecific interactive force acting on all atoms and molecules is that caused by thermal energy. This is the phenomenon responsible for Brownian motion that can be observed indirectly with light microscopy. All atoms and molecules located in a fluid, such as in liquids and gases are subjected to Brownian motion. The intensity of the Brownian movement increases with increasing temperature. The energy level of Brownian interactions at body temperature are slightly less than 1 kcal per mol.
Finally, there are the ionic associations that occur primarily in solutions. These are highly variable, weak attractions and repulsions between ions of different and like charges respectively. They vary based on the combined concentrations of all ions in a given solution.
Relative strengths of noncovalent bonds:
- hydrophobic packing, occurs in solutions of water, dipole related, very weak
- thermal energy, slightly less than one kcal per mol
- ionic interactions, very weak, vary with concentration of ions in a solution
- Van der Waals interactions, approximately 1 kcal per mol
- hydrogen bonds, 1 – 5 kcal per mol
Relative strengths of several common covalent bonds:
- hydrolysis of adenosine triphosphate (ATP) bond, 7.3 kcal per mol
- hydrolysis of phosphoester, glyceryl – 3 – phosphate bond, 2.2 kcal per mol
The Dipole Moment of Water
A unique characteristic of a water molecule is its dipole moment. This is a result of the asymmetrical configuration of the H20 molecule. The covalent bonds formed by the two hydrogen atoms with the oxygen atom are at an angle of 104.5° with one another rather than 180° required for symmetry. (See figure 3 below.) There is a slight positive charge associated with the hydrogen atoms and a slight negative charge at the oxygen end of the molecule. Because of the asymmetry, this results in a slight dipole electrical charge or moment across the molecule.
The dipole moment of water makes it an excellent solvent. For example, when sodium chloride crystals are placed in water, the sodium and chloride atoms tend to dissociate, forming ions which are positively and negatively charged respectively. Because the outer electron of sodium is only weakly attached and therefore tends to be in the possession of the chloride ion, the dipole moment of the water molecule can contribute to this process attracting the positively charged sodium ion to the negatively charged oxygen end of the water molecule while the positively charged hydrogen end of the water molecule attracts the negatively charged chloride ion. In a similar manner water is capable of dissolving many different compounds, contributing to their ionic status in solution.
The dipole moment of the water molecule also results in extensive hydrogen bond formation, both between water molecules and between water and other nonionic molecules such as glucose, particularly with the [OH+] group on the sugar molecule. Similar to salt crystals, sugar crystals readily dissolve in water by this slightly different mechanism. This phenomenon of many water molecules surrounding dissolved molecules is known as hydration or hydrophobic packing and occurs extensively in association with the many large macromolecules responsible for carrying on the complex chemical reactions that support life. They essentially all occur while dissolved in water.
Liquid water is a highly dynamic substance. Not only are the molecules in constant motion as a result of Brownian movement that is directly related to temperature, but there is constant internal motion within each water molecule in which the oxygen and hydrogen atoms are moving in reference to one another, causing slight variation in their bond lengths. There are symmetrical movements, asymmetrical movements, vibrational movements and rocking movements of the hydrogen and oxygen atoms. In general the hydrogen atom motion is much greater due to its relatively smaller size and mass. (See figure 3 below.)
The presence of water in liquid form is generally considered absolutely essential for life. In many ways it is the most remarkable compound in existence. Oxygen is absolutely essential to life as we know it. Hydrogen is by far the most abundant element in the universe. When hydrogen and oxygen are brought together they combine explosively to form water which is by far the most widely used fire retardant, as well as providing a unique environment in which the processes characteristing life can occur.
Water molecules
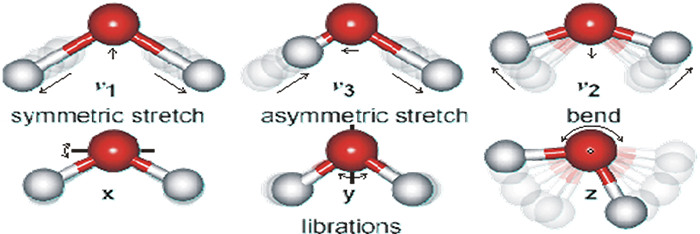
Figure 3
The Amphoteric Nature of Water
The amphoteric nature of water is its ability to act as both an acid and a base; it can donate a proton [H+] acting as an acid, and it can accept a [H+], acting as a base. Pure water is neutral. But pure water is rarely found in nature. Even rainwater, as it condenses in the atmosphere to form droplets, and falls to earth, dissolves small amounts of carbon dioxide from the air, forming weak carbonic acid.
H2O + CO2 = H2CO3
If ammonium is present in the atmosphere, it will be dissolved by falling raindrops yielding ammonium hydroxide, which is a weak base.
NH3 + H2O = NH4OH
The atoms of pure water dissociate to a small degree yielding equal amounts of hydronium ions [H3O+ or H+] and hydroxide ions, [OH-].
2 H2O = H3O++ OH-
The concentration of hydrogen ions [H+] in blood plasma is very low, measuring 0.00000004 moles per liter or 4 X 10-8 moles per liter. To avoid such an unwieldy number, the concept of pH was introduced which is defined as: pH equals minus the logarithm of the concentration of hydrogen ions, [H+], expressed in moles per liter. Thus, the concentration of hydrogen ions in plasma, expressed as pH, is equal to 7.4. The normal range of human blood plasma pH is 7.3 to 7.5; the pH range compatible with life is 6.8 to 7.8. The pH is carefully controlled primarily by the respiratory rate which controls the amount of dissolved CO2 in plasma, and the kidneys which eliminate access metabolically generated acids or bases by a complex process of glomerular filtration and renal tubular reabsorption. As a result, normal urine tends to be acidic with a pH in the range of 5 to 8. Precise control of this narrow range of the plasma pH is essential for life since an abnormal pH can result in denaturation of proteins, causing unfolding and loss of function.
Protein Synthesis
Proteins represent large molecular structures composed of many atoms of a relatively select group of elements. These form long chains of amino acids, referred to as polymers. On examination of the elemental composition of living organisms, one is struck by the surprisingly narrow selection of elements making up the great majority of the atoms in proteins. Water is by far the most prevalent compound in living organisms responsible for 80 to 90% of body weight. As a result, hydrogen is by far the most prevalent element, responsible for approximately 50% of the all atoms in living organisms, with oxygen the second most common element. The other common elements in descending order of frequency are carbon, nitrogen, phosphorus and sulfur. Additional elements occasionally encountered include calcium, potassium, iron, zinc, magnesium, manganese, fluorine, and iodine. We are all familiar with iron deficiency anemia and goiter due to iodine deficiency. The total number of essential elements in humans is 26, and for bacteria it is about 16.
Proteins are composed of varying combinations of 20 basic amino acids, nine of which are considered essential in humans. An essential amino acid is one that the human body is unable to synthesize from other protein sources and therefore must be supplied in the diet on a regular daily basis. Our bodies are capable of synthesizing the remaining 11 basic amino acids from ingested proteins. However, it is important to obtain an adequately balanced intake of the nine essential amino acids on a daily basis. Protein synthesis is dependent on an adequate supply of all of the specific amino acids required. If there are insufficient quantities of one or more of the nine essential amino acids, protein synthesis will be curtailed accordingly. A careful analysis of the living proteins indicates more than 100 different amino acids present as a result of modifications of the 20 basic amino acids by phosphorylation, glycosylation, hydroxylation, methylation, carboxylation and acetylation.
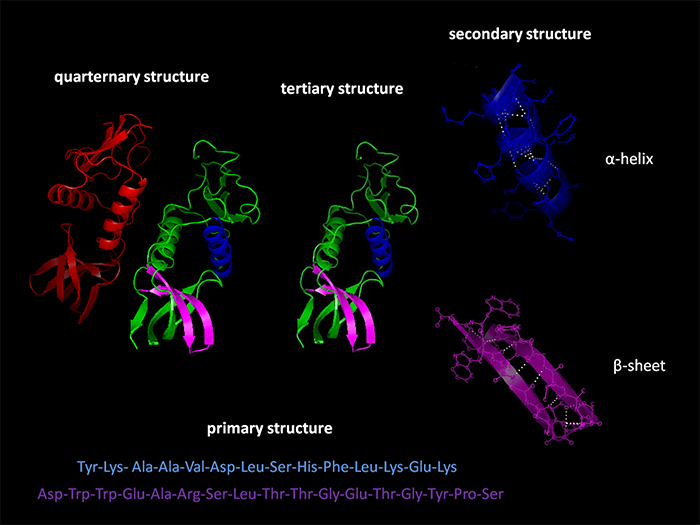
Figure 4 Courtesy of Protein Data Bank
Protein synthesis occurs in four generally recognized stages. (See Figure 4 above.) The primary stage occurs in ribosomes which are highly complex large organelles located within essentially all cells. They function as protein factories. They consist of massive complex proteins with significant amounts of RNA. They may be located within mitochondria and endoplasmic reticulum but most are located in the cytosol. Ribosomes receive directives to produce a specific protein by way of a messenger ribonucleic acid (mRNA) sent out from the cell nucleus. It is produced by transcription, in the nucleus, and represents an exact copy of a single selected short strand of DNA, except that the thymidine nucleotides in DNA, are replaced by uridine nucleotides in RNA. The mRNA provides exact information, as to the sequence of specific amino acids that need to be strung together, the number of copies to be produced, and directives as to where and how the newly produced protein should be distributed. Polymers of less than 40 amino acids in length, are usually referred to as peptides. Messenger RNA functions as a blueprint, and the ribosomes manufacture the appropriate body parts, to the exact specifications.
The secondary stage occurs as a result of various noncovalent interactions such as hydrogen bonding, ionic interactions, Van der Waals forces, and hydrophobic packing. These result in folding of the polymer into various configurations, most commonly, a random coil, an alpha helix or a beta sheet.
The tertiary stage is a continuation of the secondary in which further folding occurs as a result of further interactions of the non-covalent forces. Reversal of the tertiary stage molecular folding can be caused by higher than normal temperatures and by abnormal fluctuations of pH level. The unfolding will interfere with normal protein functioning. This process is known as denaturation and may be reversed by correcting the underlying cause.
The quaternary stage is usually performed by large complex macromolecules called chaperones, many times displaying a barrel like configuration into which tertiary proteins are inserted into the cavity where the chaperone performs specific folding maneuvers resulting in proper conformation of the molecule. The folding process may require more than one stage in which case it may be handed off to a second chaperone or co-chaperone to complete the folding process. It is well-established that identical proteins may perform entirely different functions depending on how the molecule is folded. This quaternary folding process frequently requires energy such as an ATP molecule to complete the conformation process and release of the now functional protein molecule into the cytosol.
Chaperones are usually highly complex macromolecules that have themselves undergone quaternary protein synthesis by a specialized chaperone. Thus, we have a "chicken vs egg" situation. Here we have a special form of irreducible complexity in protein synthesis that is dependent on a system where the very system that is needed must have been previously synthesized.
Intracellular Compartmentalization
Intracellular compartmentalization is a topic of great interest to cellular biologists, biochemists, and molecular biologists. It has brought about a paradigm shift in our view of cellular morphology and physiology. During the past 40 years there has been an explosion of our knowledge of intracellular processes, virtually all of which were occurring in a “black box” during Darwin's time. Our present insights into molecular activities in living cells have been made possible by a series of amazing molecular imaging and other investigative techniques that continue to advance rapidly. In 1976 X-ray imaging quickly became the major tool for examining molecular activity. This was followed in the mid 1980’s by addition of Nuclear Magnetic Resonance (NMR) imaging, and in the late 1990s by electron microscopy (EM). The rapid application of these tools to the study of cellular biology has resulted in the number of new intracellular structures discovered per year since the mid 1970’s to have grown exponentially from ten to several thousand. (14)
Fifty years ago the general concept of a living cell was that of a small packet of proteinaceous fluid, enclosed in a bi-layered phospholipid membrane in which several discrete structures such as a nucleus and several small organelles could be observed floating. We now recognize that essentially the entire interior of the cell is occupied by distinct structures performing precise functions. The concept of a cytoskeleton consisting of microtubules that provide tracks or cables on which transport motors of various types move or carry intracellular cargo from one point to another, is well-established. We now know that the cytoskeleton is an extremely dynamic structure, constantly being built up, torn down, and readjusted to accommodate intracellular activities. This condition has generated the concept of dynamic instability. Separate functional domains within the cell are associated with the specific transmembrane proteins, usually in the form alpha helixes, responsible for allowing specific moieties to enter or exit, which in most cases, is by active transport across the cell membrane. Alpha helixes are tubular shaped proteins, frequently with a central channel through which water, ions, and other small moieties can enter or exit the cell under careful control. Thus providing a conduit through which the moieties pass without contact with the phospholipids comprising the cell membrane.
It is not well understood why such dynamic changes are required constantly to maintain cellular function. It is known that prior to mitosis there is marked increased microtubular activity in preparation for spindle formation, composed of microtubules, required for separating the paired chromosomes. It has also been observed that mitochondria are constantly undergoing alterations in which short strands fuse together to produce an extensive web like pattern followed by breaking up again into short segments. This may be a means of eliminating dysfunctional mitochondria or portions thereof, representing a healing process or replacement of dysfunctional proteins.
Intracellular compartmentalization is primarily a result of the distribution of organelles within the cell. The organelles are grouped depending on their specific functions and the degree to which their functions are interrelated. The organelles are essentially all enclosed by highly specialized membranes. In most cells the smooth endoplasmic reticulum and rough endoplasmic reticulum, both consisting of extensively folded membranes, the Golgi apparatus, also a folded structure, and the early and late endosomes, are all functionally interrelated and tend to be clustered together, occupying a major compartment within the cell. The membranes of these organelles tend to be compatible, allowing protein molecules and other moieties to pass from one to another.
Ribosomes are complex macromolecules that function as protein factories and may be attached to a membranous surface such as the endoplasmic reticulum which may then serve as a storage area or distribution channel for the recently synthesized proteins. Others are associated with mitochondria. Many ribosomes however are independently located in the cytosol and release their proteins directly. A major function of the Golgi apparatus is to label and prepare newly manufactured proteins for delivery within the cell or package them for passage through the cell membrane and delivery to a distant site within the organism by way of the bloodstream.
The nucleus is usually centrally located within the cell and has large pores in its outer bi-layered membrane, allowing mRNA to pass out carrying specific instructions to the ribosomes concerning exactly what type of protein and how many copies need to be manufactured. Also, ATP, various ions, and other small moieties can pass into the nucleus through these pores.
Mitochondria also communicate directly with the cytosol, releasing manufactured ATP directly. ATP is the energy source for essentially all cellular functions. We will examine ATP synthase which is a large complex macromolecule that has been studied in great detail and represents one of the most remarkable nano-machines discovered to date. Vesicles containing specific proteins or other moieties tend to be associated with the endoplasmic reticulum where they frequently arise from the membrane wall as a budding sequence.
Other relatively independent organelles include lysosomes that are mainly concerned with dismantling and recycling intracellular material including old organelle membranes. They contain an acidic interior required to break down the proteins into peptides and amino acids. The peroxisomes break down fatty acids and play a role in neutralizing intracellular toxins. Secretory vesicles represent another unique organelle that contain secreted proteins from various sources and act as packaging material during transit to a distant site. They can fuse with the cell membrane or certain of the organelle membranes, allowing them to discharge their stored proteins. Another cell membrane structure, considered an organelle is microvilli. They represent an elegant adaptation that greatly increases the absorptive area of cell membranes. Almost the entire small intestine is lined with endothelial cells known as enterocytes on which the absorptive surface is completely covered with microvilli. The cell surface areas are literally studded with the small finger like projections, very much like the fibers of a plush carpet. They increase the absorptive surface area of the small intestine many fold.
In the past it was thought that intracellular distribution occurred primarily through passive diffusion. We now know that there is a highly specific well-organized precise method for distributing proteins and secretions. We now recognize at least three specific transport systems operating in essentially all living cells: (1) gated transport, (2) transmembrane transport, (3) vesicular transport.
- Gated transport refers to passage of molecules through the pores of the nuclear membrane simulating passing through an open gate.
- Transmembrane transport occurs primarily between the membranous organelles and surrounding cytosol via an active transport system.
- Vesicular transport is rather spectacular in that it is an active transport of vesicles, of varying sizes, carried or dragged by family of motor proteins. These are large macromolecules with paired tubular shaped projections that "walk along" microtubules. They can adjust their stride depending on the size and the cargo. These macromolecular transport motors represent another spectacular nano-machine recently discovered that is widely distributed throughout the animal kingdom.
The discovery of the above intracellular transport systems has transformed the perception of intracellular dynamics from a relatively quiet, inactive, slow-moving environment, to one that much more closely resembles a busy superhighway where cargo of various shapes and sizes are being transported hither and yon at high speeds while the very highways are simultaneously being dismantled and new ones constructed.
Macromolecular Protein Motors
There is a family of macromolecular protein motors that are widely distributed in all animal cells that are utilized extensively for intracellular transport. They primarily move vesicles, but also transport organelles and proteins within cells. The original classical studies of this phenomenon were performed on giant squid axons. The giant squid axon provides a near ideal model for experimental research on intracellular transport because it is large, 100 times the width of an average mammalian axon, and accessible. Because of its great length, cell transport plays a major role, moving proteins and other essentials such as organelles, synthesized in the cell body, to the far end of the axon.
Kenesin-1 was the initially identified and studied protein motor. It moves unidirectionally, always moving toward the positive end of the microtubule. Microtubules all have a positive and negative end. In cells, they are oriented like spokes of a wheel, radiating out from the centrosome, which functions as a microtubular organizing center (MTOC). The negative ends are centrally oriented while the positive ends are peripherally.
The Kenesin-1 motor moves in a stepwise fashion, utilizing ATP as its energy source. Its maximum speed is three microns per second or approximately 250 mm per day. (15) At this rate, it would require approximately four days to move the length of the longest human axons which extend from the neuronal cell body located in the lower spinal cord, to the nerve ending in the great toe. At maximum rate, its strides are each 16 nm. The protein motor, at maximum speed, takes an average of 375 steps per minute. That is a rapid pace by anybody's count!
Another widely recognized protein motor that is less well understood than kinesin-1, is dynein. It is a negatively oriented protein motor, thus it moves in the opposite direction on microtubules. When protein motors reach their destinations, in this case at the negative end of the microtubule, because they are unidirectional, they must be passively transported back to their starting point on the microtubule. Exactly how the protein motors are turned on and off is not understood. The motors can operate at various speeds. The speed appears to depend on the size of the cargo. For example the transportation rate of a mitochondrion is at an intermediate speed. The slowest transport rates measured are in the range of a few millimeters per day.
A third family of motor proteins is known as myosins. Of the 20 members of this family, three are of significance in humans. Myosin I is a single headed molecule associated with membranes and functions widely in endocytosis, providing the mechanical force to constrict the cell wall during cell division. Myosin V is two headed, functioning primarily in organelle transportation within cells. Myosin II is probably the most widespread and is responsible for providing the contraction force in muscles including striated muscle, smooth muscle and heart muscle. The mechanically active portion of the molecules is confined to the head and neck domains and are powered by ATP. More remarkable than the mechanical force produced by myosin activity, is the amazing neuronal control and coordination of muscle groups characteristic of all voluntary muscle function. One can hardly observed an Olympian athlete or a professional ballet dancer without experiencing a thrill of inspiration. The most amazing muscular activity is demonstrated by the human heart that starts beating at about 6 to 7 weeks gestation. The same muscle cells responsible for those earliest heartbeats will function continuously throughout the individual’s lifetime beating approximately 37 million times each year for 70, 80, 90 or possibly 100 years for a total of 2.6 to 3.7 billion heartbeats, all of which occurred with virtually no awareness or conscious control.
Structure and Function of a Basic Macromolecular Nano-machine: ATP Synthase
ATP Synthase: schematic drawing
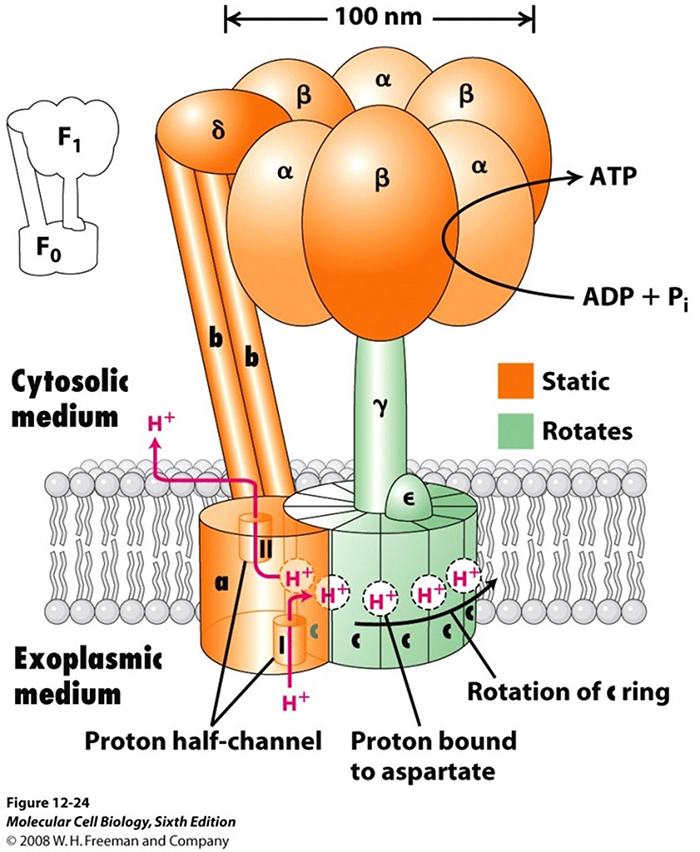
Figure 5, Courtesy of Protein Data Bank
ATP Synthase: Macromolecule
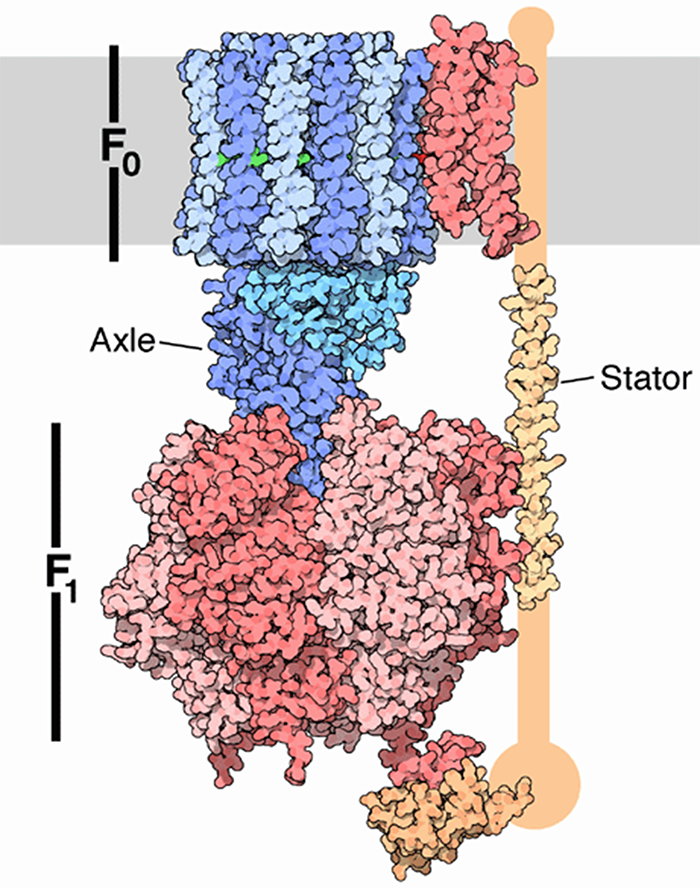
Figure 6, Courtesy of Protein Data Bank
(Note: the orientation of figure 6 is reversed.)
ATP synthase is a widely distributed macromolecular nano-machine located in mitochondria. (See figures 5 and 6.) It is the primary generator of ATP throughout the entire animal kingdom. It consists of two large multi-protein components one being static, colored orange, and anchored in the mitochondrial membrane, and the other mobile, colored green, and rotating much as an armature of a motor.
The mobile component is a complex multi-protein consisting of 12 (in humans) identical, pie shaped molecules, arranged in a cylindrical configuration. It has a column like protein structure rigidly attached in a vertical manner so that when the cylindrical structure, acting as an armature rotates, the column turns with it like a driveshaft. The cylindrical shaped portion is positioned in the plane of the mitochondrial membrane. Each of the pie shaped protein structures is labeled c. Its rotation is powered by protons flowing, one at a time, along a pathway consisting of two half channels, leading from the exoplasmic medium through the mitochondrial wall and emptying into the interior cytosolic medium. Since one proton is required to rotate the armature by one c unit, in humans, twelve protons are required to produce one complete rotation.
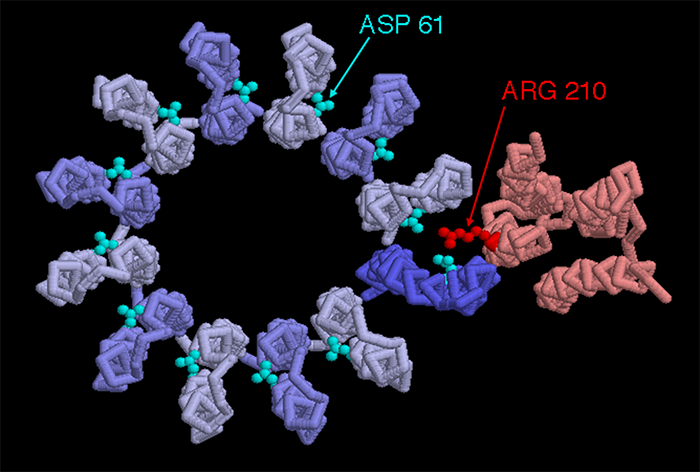
Figure 7, (Courtesy of Protein Data Bank.) ATP Synthase, FO structure, top view, showing the 210 position on the arginine amino acid and 61 position on aspartic amino acid. These represent the binding sites.
As a proton flows through the first half channel, at approximately midway through the wall, it interacts with Asp (aspartic acid) 61 on its un-protonated binding site, resulting in balancing a negative charge on this same side chain of the amino acid. It is also partially balanced by a positive charge on nearby Arg (arginine) 210. “The proton fills the empty proton-binding site and simultaneously displaces the Arg 210 side chain, which swings over to the filled proton binding site on the adjacent c subunit. As a consequence the proton bound at that adjacent site is displaced. The displaced adjacent proton moves through half channel II and is released into the cytosolic space leaving an empty proton-binding site on Asp 61. Counterclockwise rotation of the entire c ring moves the "empty" c subunit over half channel I.”(16) Rotation is caused by thermal/Brownian motion, and this is all powered by "proton – motive force" across the membrane that drives the flow of protons through the membrane, from the exoplasmic to the cytosolic medium.
As mentioned earlier, there is a static component which is actually larger in over-all size than the rotating component. It measures slightly more than 100 nm in diameter. It is firmly anchored in the mitochondrial membrane. There are two large paired linear structures with an additional intervening protein macromolecule that support a large donut shaped structure, consisting of three pairs of alternating macromolecules. They sit on top the rotor shaft, described earlier, much like the head of a mushroom sits on its stalk.
The three pairs of large macromolecules, labeled alpha and beta, make up the mushroom head-like structure, each occupies 120° of the 360o ring. There is an asymmetrical molecule attached firmly to the upper end of the rotating shaft. This structure functions as a cam. As the cam rotates each 120° segment of rotation it makes intimate contact with the binding sites in each of the three paired macromolecules, where it causes a change in the conformational state in each. There is an actual change in the shape of the protein structures causing a mechanical shift at each of the binding sites.
As it rotates, it cycles through each of three stages at the binding sites. The first stage is the releasing stage of a molecule of ATP; the second stage is weakly attracting a molecule of ADP + Pi; and the third stage is strongly attracting the ADP + Pi resulting in a firmly bound molecule of ATP, ready for release into the cytosolic medium next cycle.
The rate of rotation has been experimentally measured at approximately 134 revolutions per second. The rate of generation of ATP molecules has been experimentally measured at about 400 ATP molecules per second. Since three molecules of ATP are produced and released into the cytosolic medium with each rotation, these experimental values are in excellent agreement.
Well-recognized feedback systems controlling the rate of ATP synthesis are recognized such as the concentration of ADP. There is also a coupling of oxidation of NADH and FADH2 to the synthesis of ATP, so if the resulting proton–motive force is not dissipated during the synthesis of ATP, the transmembrane gradient resistance will increase and eventually block further reaction.
In summary, ATP synthase is a large macromolecular nano-machine that performs one of the most critical functions in the bacteria, animal and plant kingdoms. It is an essential component for almost all living cells. It generates the fuel required to power all of the major physiological functions of living organisms.
It consists of twenty-five distinct macromolecules in which five are singular, four are paired and one consists of twelve identical copies arranged in a cylindrical shaped complex protein acting like an armature of a motor that rotates at an experimentally determined rate of 134 revolutions per second. (8040 revolutions per minute). It is uniquely situated in the wall of mitochondria and provides a pathway for protons to flow across the membrane, traversing two, one-half, uniquely arranged channels, driven by the proton – motive force. (16)
It is estimated that in humans, the amount of ATP produced and utilized each day is approximately equal to one's total body weight. Without ATP all animal life would immediately cease. In fact the sodium–potassium pump, responsible for maintaining the relatively negative concentration of sodium and relatively positive concentration of potassium within all cells, a condition essential to all living cells, is totally dependent on ATP as its source of energy. How is it possible to conceive that an irreducibly complex macromolecular nano-machine, such as ATP synthase, that is absolutely essential to life, could possibly evolve when the evolutionary process based on random mutations and survival of the fittest, is dependent on the very life processes that could not occur without this most remarkable nano-machine?
The highly accurate animated video depicting intracellular molecular activity, produced at Harvard and distributed on YouTube, entitled “Inner Life of the Cell” is recommended as a sequel to this presentation. It is located at: https://www.youtube.com/watch?v=wJyUtbn0O5Y
Conclusion
What characterizes living molecules? The elements are no different whether they compose an organic or inorganic molecule. They appear identical and interchangeable. The obvious characteristic of elements composing living molecules is that they perform functions in an organized fashion. One's digestive enzymes, for example, dismantle ingested food into absorbable component parts that are needed to maintain life. These are then absorbed, processed, stored and distributed as needed. Muscle cells participate in movement and locomotion; myocardial cells pump blood day and night year after year; red blood cells transport oxygen and carbon dioxide; neurons transmit neural signals to extremity muscles to induce locomotion; highly specialized retinal cells transmit visual data along optical neural pathways to the occipital cortex where visual phenomena are interpreted; other neural centers of the brain enable self-consciousness, other consciousness and even contemplation of cosmic consciousness. Because of organization, so many moving parts seem to be synchronized, so living organisms can function effectively. From whence is this organization? How did the incredible density of information become established in the first DNA molecules? How did a cell arise before there were cells?
Given our recently greatly expanded understanding of the complexities of intracellular molecular activity, one must reasonably ask, are there any cells that are not irreducibly complex? This is not a disparagement of Charles Darwin's brilliant observations concerning the interrelatedness of all living organisms nor are we questioning the fact of evolution. He could not have imagined the intracellular complexities that we have recently uncovered. During his time it occurred totally in a "black box."
Where did the apparent sense of purpose that characterizes so many living organisms come from, the drive to obtain food, to grow, to reproduce, to avoid predators and protect one's young? How did the concept of a genetic code arise spontaneously? Encoding information is dependent on high-level cognitive thinking.
How did the first cell acquire ATP to run its sodium-potassium pump that generates and maintains the transmembrane electrical potential that is an absolutely essential component of every living cell? ATP synthase, responsible for generating ATP required to power most intra-cellular functions, depends on the proton gradient across the mitochondrial membrane for its energy source as well as the electron transport cascade that produces the proton – motive force which supplies energy to generate ATP. How did the large complex macromolecular ribosomes form when there were no functioning protein factories to produce the polypeptides of which they consist?
Our recently acquired ability to analyze living organisms at the molecular level has opened our eyes to myriads of nano-machines that function at unbelievably high speeds, performing precise actions, with astounding accuracy, based on specific instructions, coming from encoded sources stored in chemical libraries, that have reliably preserved and transmitted the mysteries of life from generation to generation for hundreds of millions of years
There are so many moving parts! How do they avoid colliding? They seem so correlated and synchronized.
In looking back, we observe a gigantic biological Big Bang about 520 million years ago. Suddenly many of the phyla and complex multicellular body plans, burst onto the seascape. The apparently explosive creative forces, during the Cambrian Period have since subsided. The tempo suddenly changed and stamina and ingenuity emerged. A long period of sorting, sifting, and diversification followed that still continues. Diverse life forms are to be found with every searching glance.
Now that we Homo sapiens have finally emerged at the top of the food chain, and no longer need to expend every waking hour seeking nourishment and providing for protection, we have time to think and look around us. We have extended our vision significantly both inwardly and outwardly. What we are now seeing in both directions is absolutely astounding.
At the beginning of my career we struggled to identify intracellular structures with the light microscope that resolved objects down to approximately 1 µm. The electron microscope soon extended our resolution by several magnitudes, to resolutions of a few angstroms. We observed living molecules measured in nanometers. Individual eukaryote cells that once were thought to be small packages of proteinaceous fluid wrapped in phospholipid pouches, are in fact complex factories filled with millions of nano-machines rushing about, performing at unbelievable speeds, never taking breaks, and performing all maintenance, repair, including replacement with new machines, on-the-fly. And if this factory happens to be an enterocyte located in the small intestine, the whole factory will be replaced, on average, every five days.
How many cells, each containing thousands of nano machines, are there in the average human body requiring organization and supervision? Is it 10 trillion or 100 trillion? Both figures appear in print. We really do not know, but which ever it is, it is beyond human comprehension. And to further complicate things, these factories are not all the same. Each organ has very distinctive functions. Red blood cells carry oxygen from the lungs to the tissues and carbon dioxide from body tissues back to the lungs. White blood cells fight infections, clear inflammation, and manufacture antibodies against invading viruses. Specialized cells in the Islets of Langerhans in the pancreas produce insulin, liver cells produce bile, thyroid cells produce thyroxine, heart cells contract and expand pumping blood, and neurons interpret visual input, signal legs to walk, and cerebral cortical neurons to think. The brain for example has hundreds of different subtypes of neurons.
So many moving parts! Flagella rotating at 100,000 revolutions per minute, ATP synthase motor turning at 8000 RPM, producing ATP molecules at 20,000 per minute, and protein motors carrying cargo at 8 nm per pace at 375 paces per minute.
So many moving parts and moving so fast! One is reminded of an experience attending a ballet. So much movement but all perfectly synchronized to the music, all coordinated and fine-tuned. Very demanding of stamina and endurance, yet, no collisions, falls, or interruptions.
Where is the choreographer?
End Notes
1. Lodish, H, et.al. Molecular Cell Biology , W H Freeman and Company, New York NY (2013) pp.544 – 552.
2. Savage, D F, et al., "Architecture and Selectivity in Aquaporins: 2.5 Å X-Ray Structure of Aquaporin Z," PLoS Biology 1, no. 3 (December 22, 2003):doi: 10.1371/journal.pbio.
3. Shapiro, L, Losick, R, "Protein Localization and Cell Fate In Bacteria," Science 276 (May 2, 1997): 712 – 18.
4. Harold, F M, The Way of the Cell, Oxford University Press, Oxford, England (2001) pp. 65-66.
5. Lili, C, "Traditional Theory of Evolution Challenge," Beijing Review (31 March – 6 April 1977): 10.
6. Levinton, J, "The Big Bang of Animal Evolution," Scientific American, (November 1992): 84 – 91; Kerr, R A, "Evolution’s Big Bang Gets Even More Explosive," Science 621 (1993): 1274 – 75; Monastersky, R, "Siberian Rocks Clock Biological Big Bang" Science News 144 (4 September 1993): 148.
7. Gould, S J, Wonderful Life, The Burgess Shale and the Nature of History W. W. Norton and Company, Inc. New York, N Y (1989) p. 56.
8. Chen, J Y, Li, C W, Chien, P, Zhou, and Gao, F, “Weng’an Biota-A Light Casting on the Precambrian World” paper presented to the origin of animal body plans and their fossil records, China 20 – 26, June 1999.
9. Lili, C, Chen, J Y, Zhou, G Q, Zhu, M Y, Yeh, K Y, The Chengjiang Biota: A Unique Window of the Cambrian Explosion, volume 10.
10. Gearty, W. "The Cambrian Explosion: Evolution’s Big Bang," Yale Scientific, 23:37 (December 23, 2013).
11. Lee, M.S.Y., et al., "Rates of Phenotypic and Genetic Evolution during the Cambrian Explosion, Current Biology, 23: 19, (October 7, 2013) pp. 1889 – 1895.
12. Gould, S J, Wonderful Life, The Burgess Shale and the Nature of History W. W. Norton and Company, Inc. New York, N Y (1989) p.57
13. Lodish, H, et.al, Molecular Cell Biology, W H Freedman and Company, New York, NY (2013) page 988 – 89.
14. Krimm, Samuel; Bandekar, J. (1986). "Vibrational Spectroscopy and Confirmation of Peptides, Polypeptides, and Proteins". Advances in Protein Chemistry. Advances in Protein Chemistry 38 (C): 181 – 364.
15. Lodish, H, et.al, Molecular Cell Biology, W H Freeman and Company, New York, NY (2013) p. 833.
16. Lodish, H, et.al, Molecular Cell Biology, W H Freedman and Company, New York, NY (2013) p. 547.
Study the Book
- FAQs
- A Strategy and Practice for In-depth Study Groups of The Urantia Book
- A Study of the Master Universe
- Adopted Corrections to the 1955 Text
- An Artist's Conception of the Master Universe
- Appendices to A Study of the Master Universe
- Bible Study
- Bill Sadler Talks
- Concerning Human Survival
- Consideration of Some Criticisms of The Urantia Book
- Foreword and Part 1
- General Organization of The Urantia Book
- Guide to Pronunciation of Names and Words in The Urantia Book
- Jesus
- In His Steps - Maps of Jesus' Travels
- Index of The Urantia Book
- Jesus' Travels - Google Earth
- Publications
- Science in The Urantia Book
- Terminology
- Term List for The Urantia Book
- The Unceasing Campaign of the Master Seraphim
- The Urantia Book Concordance
- The Value of Study Groups
- Theology of The Urantia Book
- Topical Studies
- Worship and Wisdom
- Inspiring Questions
Seminars & Presentations
- The Application of Animated Whiteboard Videos
- The Atom
- A Call for a New Paradigm of Education Within the Urantia Community
- The Cosmogenetic Principle
- Crystallization of Water Vapor: What May We Discern From a Snowflake
- Implications of Free Will in the Cosmos
- Institutionalizing Education to Support the Preparation of Teachers of the Urantia Revelation
- Is There Design in Nature?
- Our Educational Imperatives
- Preparing for Spontaneity
- Putting New Wine into Fresh Wine Skins
- The Revelation as a Personal Ministry
- Safe and True
- The Seven Adjutant Mind Spirits - a Revelation for a Scientific Explanation of Mind
- Sharing the Teachings Without Name and Number
- Sharing Urantia Book Teachings - Attitude Matters!
- The Spiritual Brain
- Unicellular Organisms – The Cambrian Explosion – Nano Machines